Development of IVD Software
We support you in all areas
to web and desktop applications to mobile applications, we implement powerful, secure, and standards-compliant software for modern diagnostic systems.

Guide
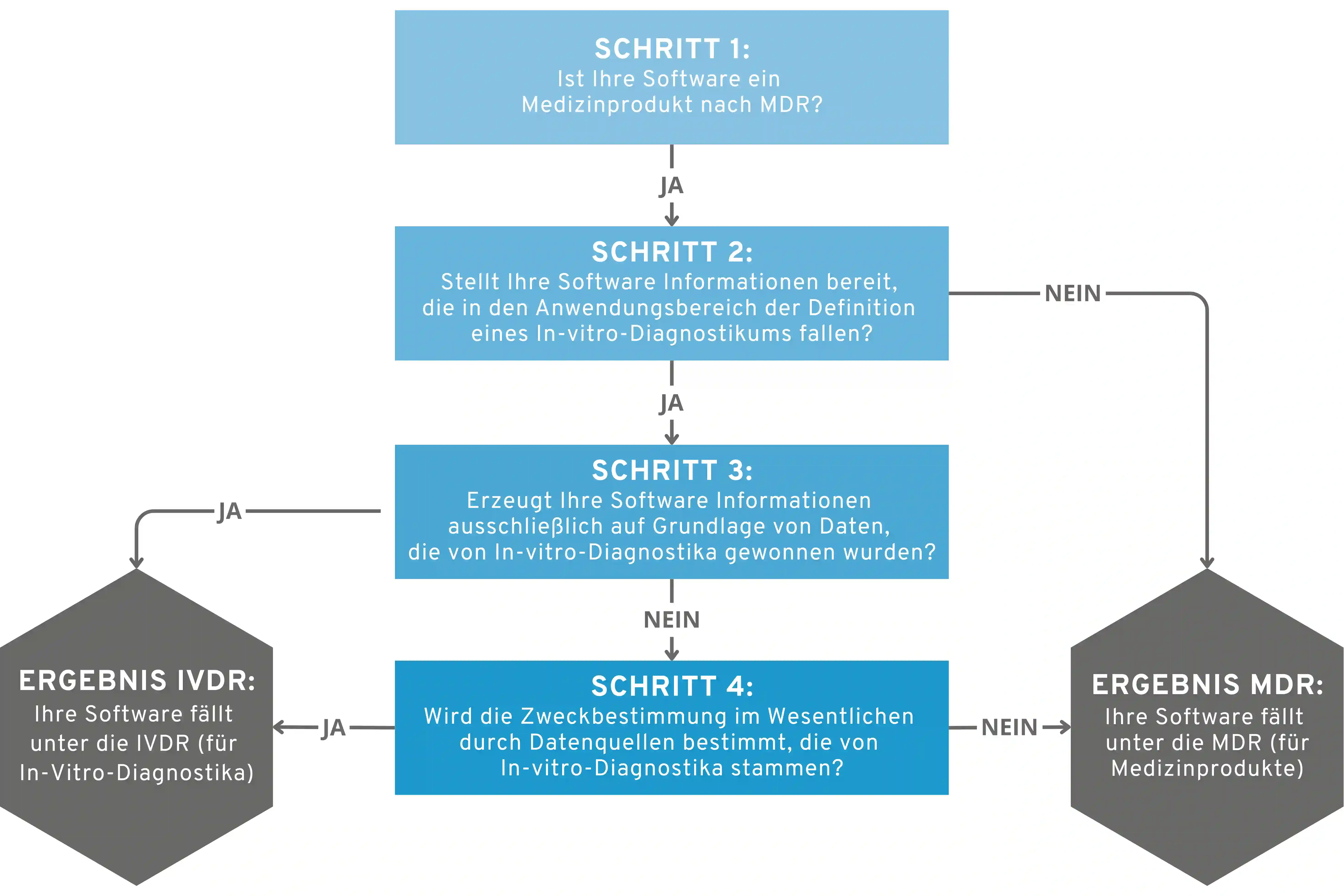
IVDR Software: Qualification & Classification
In our guide IVDR Software – Qualification and Classification, we explain…
- …when software falls under the In Vitro Diagnostic Regulation (IVDR)
- …how the qualification works
- …and according to which criteria the risk classification is carried out.
Our Expertise
Software Development
- Mobile Apps
- Web Applications
- Desktop Applications
- Backend Components
- Embedded Systems
Information Security
-
Certified Cyber Security according to ISO 27001, IEC 81001-5-1, MDCG 2019-16
-
Penetration Tests according to OWASP & BSI TR-03161
-
Information Security Management
-
GDPR Compliance
Technical Documentation according to IVDR
- Quality Management (ISO 13485)
- Software Lifecycle Processes (IEC 62304)
- Risk Management (ISO 14971)
- Usability Engineering (IEC 62366)
Regulatory Consulting
- Approval as an in vitro diagnostic device according to IVDR
- Classification of in vitro diagnostic medical devices
- Identification and Implementation of IVDR Requirements
- Validation of IVD Products
Our Process

1. Conception
- Requirements Analysis
- Definition of the Intended Purpose
- Planning of the System Architecture
- Selection of suitable Technologies
- Determination of Regulatory Strategy

2. Planning
- Project Planning
- Definition of the Project Team
- Clarification of Responsibilities
- Identification of Requirements (technical and regulatory)
- Prioritization of Functions
- Risk Analysis & Planning of Control Measures (IT security and patient safety)

3. Development & Documentation
- Technical Implementation of Requirements
- Agile Development of IVD Software
- Compliance with IVDR Requirements
- Regulatory Compliance of Documentation
- Product Labeling

4. Verification & Validation
- Software Tests
- Clinical Evaluation and Testing
- Penetration Testing
- Safety Inspections

5. Approval as an In Vitro Diagnostic Device
- Generation of a UDI Number
- Product Registration (EUDAMED)
- Support for the Audit by Notified Bodies

6. After Approval
- User Support
- Maintenance of the IVD Software
- Post-market Surveillance (PMS)
- Post-market Clinical Follow-up (PMCF)
IVD Software Types
We develop various types of software solutions for in vitro diagnostic medical devices (IVDR) that are tailored to your specific requirements and area of application. Whether you need standalone software, embedded software or control software for devices, we offer customized solutions that guarantee the highest quality and compliance.

Standalone Software
Standalone software solutions are independent applications that operate independently of other systems. This type of software is often used in clinical environments to support diagnoses or enable analysis processes. We ensure that this software is not only user-friendly and reliable, but also meets all regulatory requirements of the IVDR.

Embedded Software
For IVDR devices that work with integrated software, we offer customized embedded solutions. This software is integrated as part of an in vitro diagnostic device and implements the core functionality. For more information about our embedded software services, please visit our Embedded Software Service page.

Control Software for Devices
Control software is responsible for the direct control and operation of IVD devices. It ensures that the device works reliably and that all diagnostic processes are carried out correctly. We guarantee full compliance with IVDR requirements so that your devices can be used safely and efficiently.
What makes us special

ISO 13485 certified
Our quality management system is certified according to ISO 13485. This ensures that we develop software for in vitro diagnostics in compliance with regulations and meet the requirements of the IVDR for quality management systems.

External placing on the market
Upon request, we can assume the role of legal manufacturer for your IVDR software product. We bear legal responsibility for compliance with all regulatory requirements of the IVDR, allowing you to concentrate on your core competencies such as sales and marketing.

ISO 27001 certified
QuickBird Medical is ISO 27001 certified for information security. We are experts in cybersecurity and data protection, especially for health data.
Are you planning to develop a Software Product for In Vitro Diagnostics?
Our team will support you in the planning and technical implementation of your IVD software. Benefit from our experience in implementing the regulatory requirements of the In Vitro Diagnostic Medical Devices Regulation (IVDR). Contact us to arrange a no-obligation initial consultation. Together, we will find out how we can support you in your project.