Development of Medical Software, Health Apps and DTx
In the heart of Munich, we develop mobile apps, web applications and cloud systems for companies in the healthcare sector.
You are planning the implementation of a Medical Software or DTx? In close collaboration with you, we take care of the conception, design and technical implementation of your software idea – certified according to ISO 13485 and ISO 27001.
Our Services
DIGITAL HEALTH APPLICATIONS (DIGA)
We develop your DiGA in compliance with all regulatory requirements and support you on your way into the DiGA directory.
MEDICAL DEVICE SOFTWARE (SAMD)
We develop your software medical device according to MDR and FDA requirements (21 CFR) – certified with ISO 13485 & ISO 27001.
HEALTH SOFTWARE & APPS
We develop your mobile app or web application that helps people to lead a healthier and better life in the long term.
LEGAL MANUFACTURER SERVICE
Certify a medical device without regulatory effort? We assume legal manufacturer responsibility for your medical device software or DiGA.
DECISION SUPPORT SYSTEMS
We develop customized software systems for your company that support specialists in the diagnosis or treatment of diseases.
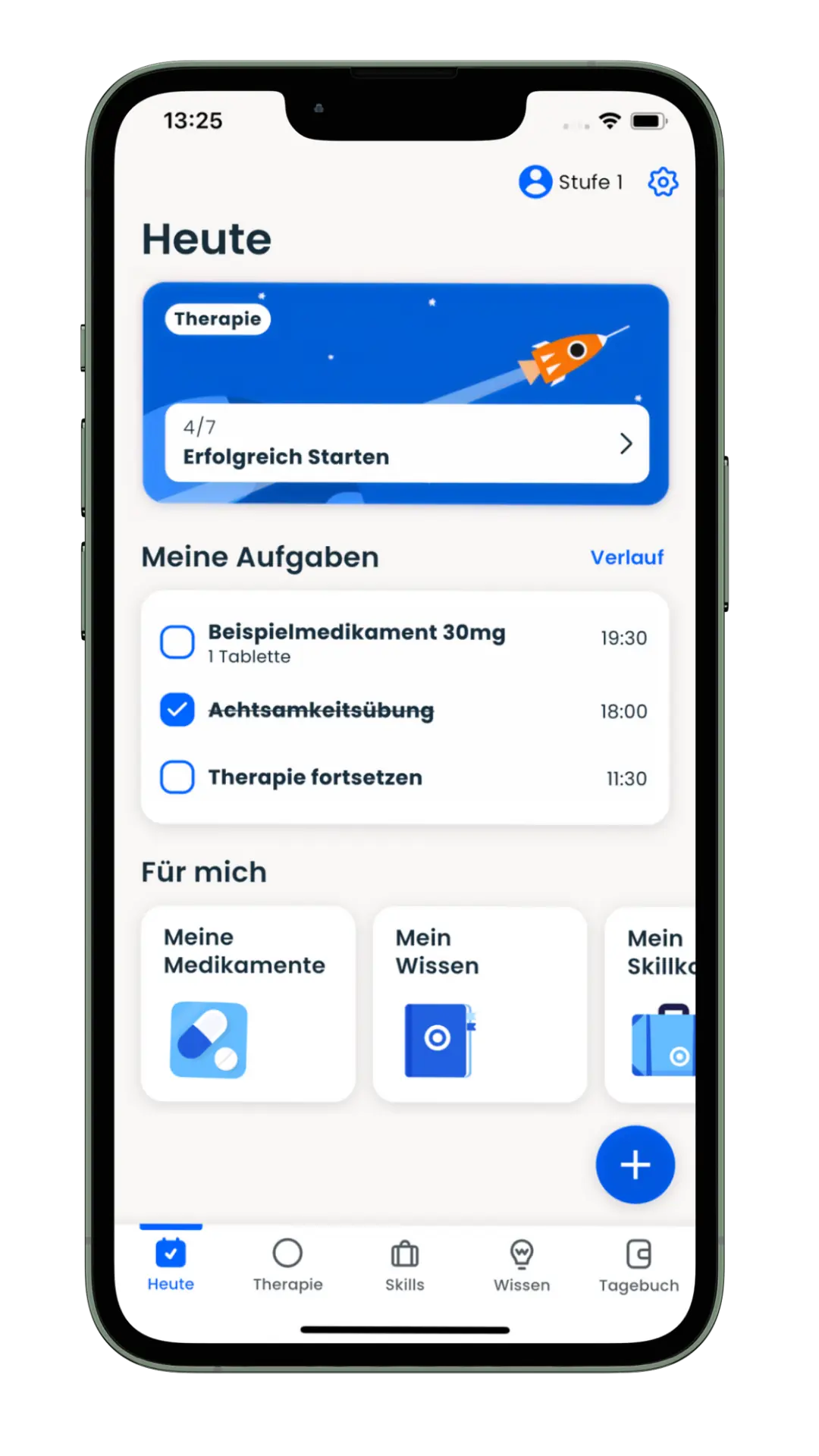
ORIKO®
DiGA that measurably help patients
We develop digital health applications (DiGA) for pharmaceutical companies such as Takeda and start-ups such as MiNDNET Solutions. The ADHD therapy app ORIKO® is the first DiGA for adults with ADHD to be listed in the BfArM directory.
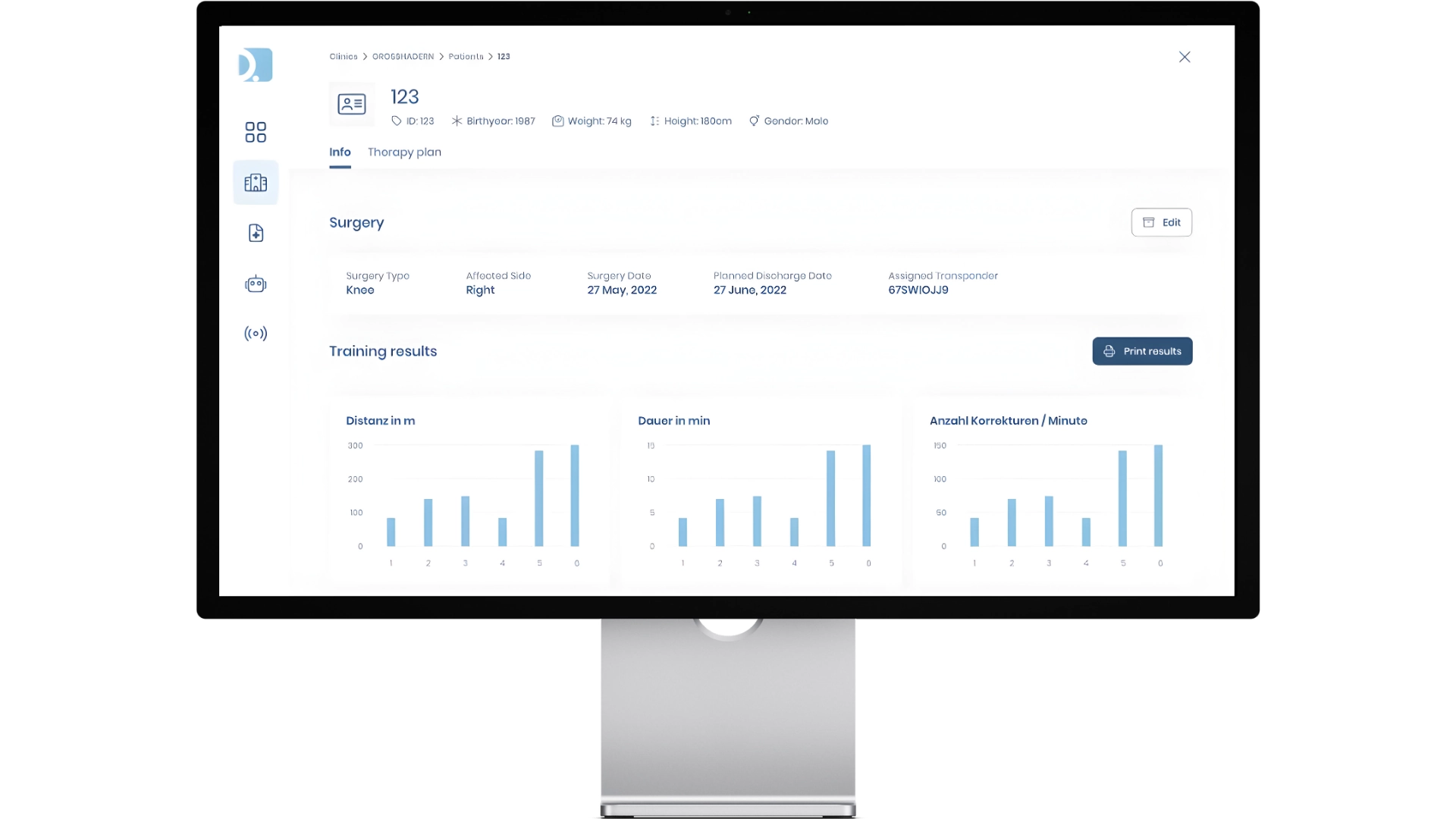
TEDIRO
Software systems that relieve hospital staff
For companies such as TEDIRO, we develop software solutions that enable specialist staff in clinics to work more efficiently. The Therapy Management System developed by QuickBird Medical automates the planning, scheduling, and implementation of groundbreaking gait training with robots.
Our Process
Strategy & Feasibility
Together with you, we develop a suitable product strategy, the business model and the basic concept of the software. We identify the opportunities and risks of various solutions and provide you with comprehensive advice.
Concept and Design
We create a design concept based on your ideas, all regulatory requirements and user needs. Design work is integrated into the product development cycle in an agile manner and continuously optimized.
Implementation and Testing
In an agile process, we develop a software product for you that is ready for use by patients and HCPs. We ensure that your users can rely on the product and that data security is covered on the basis of BSI guidelines.
Maintenance & Support
Of course, we won’t let you down even after your product has been published. Our team remains at your side to maintain your software on an ongoing basis, operate your server and continuously integrate new features according to your individual requirements.
Our Motivation
Whether we live a satisfied life depends on many factors. However, the foundation in any case is our physical and mental health. We want to contribute to the improvement of human health by developing medical software and health apps for medical technology companies, pharmaceutical companies and clinics. The common goal with our customers:
Enabling people to live healthier lives.

What makes us special

ISO 13485 certified
Our quality management system is certified according to ISO 13485. This ensures that we develop software as a medical device in compliance with regulations and meet the requirements of the MDR and FDA (21 CFR) for quality management systems.

ISO 27001 certified
QuickBird Medical is ISO 27001 certified for information security. We are experts in the areas of cybersecurity and health data protection.
Are you planning to implement medical software
or DiGA?
Contact us for a free initial consultation. We will give you an estimate of the effort and timeframe required to implement your project. We will also examine the regulatory and strategic framework conditions for your product.